When you hear the term HER2-positive breast cancer, it is a distinct molecular subtype of breast cancer characterized by overexpression of the human epidermal growth factor receptor 2 protein on cancer cell surfaces. This specific classification affects roughly 15% to 20% of all breast cancer patients. For years, this was considered one of the most aggressive forms of the disease. But thanks to major breakthroughs in oncology, the outlook has changed dramatically. The discovery of HER2 as an actionable therapeutic target about 25 years ago led to the approval of trastuzumab (Herceptin), which is the first HER2-targeted monoclonal antibody approved for treating HER2-positive breast cancer. Today, we have a whole arsenal of drugs designed specifically to stop these cells from growing and spreading.
Understanding your treatment options can feel overwhelming. There are different classes of drugs, each with its own way of working, side effects, and place in your treatment plan. This guide breaks down exactly how these targeted therapies work, what to expect during treatment, and why certain drugs are chosen for specific stages of the disease.
How HER2 Targeted Therapies Work
To understand these treatments, you first need to know what HER2 is. It’s a protein that sits on the surface of some breast cancer cells. In HER2-positive cases, there are too many of these receptors. They act like gas pedals, constantly telling the cancer cells to grow and divide. Standard chemotherapy attacks all fast-growing cells, but targeted therapies are more precise. They go after the HER2 receptors directly.
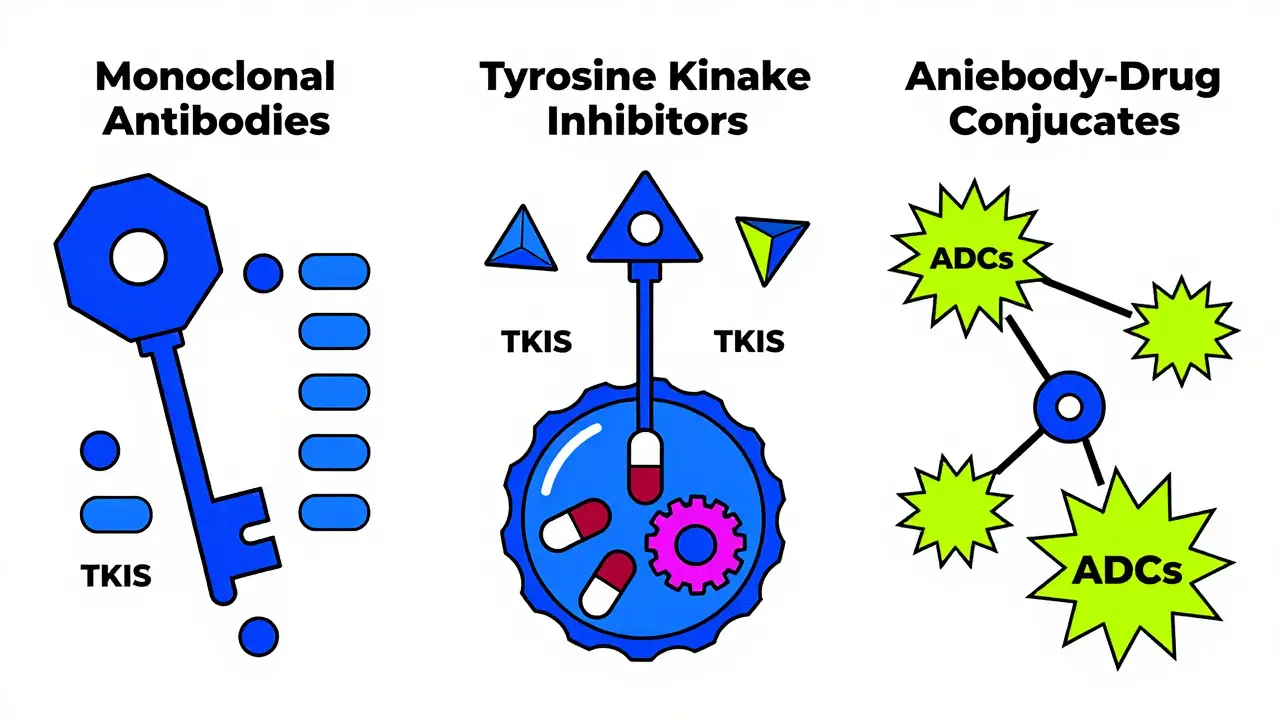
These therapies generally fall into three main categories:
- Monoclonal Antibodies: These are lab-made proteins that bind to the HER2 receptor. By attaching themselves, they block the signals that tell the cancer to grow. They also flag the cancer cells so your immune system can find and destroy them.
- Tyrosine Kinase Inhibitors (TKIs): These are smaller pills that get inside the cancer cell. They block the enzymes inside the cell that process the growth signals, effectively cutting off the communication line.
- Antibody-Drug Conjugates (ADCs): Think of these as "smart bombs." They use an antibody to find the HER2-positive cell, then deliver a powerful chemotherapy drug directly inside it. This limits damage to healthy cells.
The goal is always the same: stop the tumor from getting bigger and prevent it from spreading to other parts of your body. Combining these targeted drugs with surgery, radiation, or traditional chemotherapy often gives the best results.
The Backbone: Monoclonal Antibodies
Trastuzumab remains the foundation of almost every HER2-positive treatment plan. It’s been around since 1998, and it works incredibly well for many people. You usually receive it through an IV infusion or a subcutaneous injection. Doctors typically prescribe it for one year in early-stage breast cancer.
Often, doctors pair trastuzumab with another antibody called pertuzumab (Perjeta). Pertuzumab targets a different part of the HER2 molecule. Together, they provide "dual blockade," which is much more effective at stopping tumor growth than using just one. For convenience, there is also a fixed-dose combination called Phesgo, which contains both pertuzumab and trastuzumab in a single subcutaneous injection. Patients love this because it turns a 90-minute IV infusion into an 8-minute shot.
If the first lines of defense don’t work, there are newer options. Margetuximab (Margenza) is a monoclonal antibody used when other HER2 therapies have stopped working. It’s designed to engage the immune system more aggressively against the cancer cells.
Smart Bombs: Antibody-Drug Conjugates (ADCs)
ADCs represent a huge leap forward in precision medicine. Instead of flooding your whole body with chemo, these drugs carry the poison only to the cells that have HER2 on them.
The first widely used ADC is ado-trastuzumab emtansine (T-DM1/Kadcyla). It links trastuzumab to a chemotherapy agent called DM1. It’s commonly used if cancer comes back after initial treatment.
More recently, trastuzumab deruxtecan (T-DXd/Enhertu) has become a game-changer. Clinical trials showed it significantly outperformed T-DM1. In fact, it reduced the risk of disease progression or death by 72% compared to older treatments. What’s even more exciting is that T-DXd works not just for HER2-positive cancer, but also for "HER2-low" breast cancer. This expands treatment options for millions of women who previously didn’t qualify for targeted therapy.
However, ADCs come with serious risks. T-DXd carries a boxed warning for interstitial lung disease (ILD), a type of pneumonia-like inflammation in the lungs. About 10-15% of patients in trials experienced this. You’ll need regular monitoring and must report any new cough or shortness of breath immediately.
Pill Power: Tyrosine Kinase Inhibitors (TKIs)
Unlike injections, TKIs are oral medications. They’re small molecules that can enter cells easily. This makes them particularly useful for reaching places other drugs can’t, like the brain.
Tucatinib (Tukysa) is a standout here. It was the first small molecule inhibitor proven to be effective against brain metastases in HER2-positive breast cancer. Studies showed it improved progression-free survival significantly compared to placebo. If you have cancer that has spread to your brain, tucatinib combined with trastuzumab and capecitabine is often the go-to regimen.
Other TKIs include lapatinib (Tykerb) and neratinib (Nerlynx). Neratinib is often used as extended adjuvant therapy to lower the risk of recurrence after you’ve finished your main treatment.
The biggest downside of TKIs? Gastrointestinal side effects. Diarrhea is extremely common, especially with neratinib. Many patients report severe diarrhea that forces them to stop the medication. Doctors usually prescribe prophylactic loperamide (Imodium) to manage this, starting before symptoms even appear.
| Drug Name | Type | Administration | Key Benefit | Main Side Effect |
|---|---|---|---|---|
| Trastuzumab (Herceptin) | Monoclonal Antibody | IV or Subcutaneous | Backbone of treatment; long safety record | Cardiotoxicity (heart failure) |
| Pertuzumab (Perjeta) | Monoclonal Antibody | IV or Subcutaneous | Dual blockade synergy with trastuzumab | Diarrhea, infusion reactions |
| T-DXd (Enhertu) | Antibody-Drug Conjugate | IV Infusion | High efficacy; works for HER2-low | Interstitial Lung Disease (ILD) |
| Tucatinib (Tukysa) | Tyrosine Kinase Inhibitor | Oral Pill | Effective against brain metastases | Diarrhea, liver enzyme elevation |
| Neratinib (Nerlynx) | Tyrosine Kinase Inhibitor | Oral Pill | Reduces recurrence risk in adjuvant setting | Severe diarrhea |
Managing Side Effects: Heart and Lungs
Targeted therapies are easier on the body than traditional chemo-you likely won’t lose your hair or suffer severe nausea. But they do have unique risks that require careful monitoring.
Heart Health: Trastuzumab and pertuzumab can weaken the heart muscle. About 2-7% of patients develop symptomatic heart failure. Before starting treatment, your doctor will check your left ventricular ejection fraction (LVEF) via an echocardiogram or MUGA scan. You’ll repeat this test every 3 months during treatment. If your heart function drops, your oncologist may pause the drug or add heart-protective medications.
Lung Health: As mentioned, T-DXd can cause ILD. Symptoms include dry cough, fever, and shortness of breath. Because these can mimic flu symptoms, it’s crucial to tell your care team immediately if you feel unwell. Early detection allows doctors to treat it with steroids before it becomes life-threatening.
Gut Health: For TKI users, diarrhea is the enemy. Don’t wait until it’s bad to act. Take loperamide at the first sign of loose stool. Stay hydrated, eat bland foods, and avoid dairy if it worsens things. Your doctor might adjust the dose if it doesn’t improve.
Treatment Sequencing: What Comes First?
Your treatment path depends heavily on whether your cancer is early-stage or metastatic.
Early-Stage (Adjuvant/Neoadjuvant): If you have larger tumors (>2 cm), doctors often start with neoadjuvant therapy (before surgery). The standard is dual HER2 blockade: trastuzumab plus pertuzumab, along with chemotherapy. After surgery, you continue trastuzumab to complete one year total. Some patients with high-risk features may take neratinib afterward to further reduce recurrence risk.
Metastatic (Advanced): The sequence is more flexible. First-line treatment is usually trastuzumab + pertuzumab + a taxane chemotherapy. If the cancer progresses, second-line therapy often involves T-DM1. Third-line and beyond now frequently feature T-DXd, especially if you have HER2-low status. Tucatinib combinations are reserved for those with brain metastases or who haven’t responded to prior ADCs.
The landscape is moving fast. New drugs are entering clinical trials every month. Bispecific antibodies and next-generation ADCs aim to overcome resistance and improve safety. Always ask your oncologist about clinical trials-they might offer access to cutting-edge treatments not yet available to the public.
Living with Treatment: Patient Perspectives
Reading medical guidelines is one thing; living with the treatment is another. Patients share real-world experiences that help set expectations.
Many praise the convenience of subcutaneous formulations. One patient noted, "The 5-minute injection versus 90-minute infusions has been life-changing for my treatment schedule." It means less time at the clinic and more time at home.
Others struggle with anxiety. Regular heart scans create stress. "The worry of wondering if treatment will have to stop due to heart issues" is a common sentiment. Open communication with your cardiologist and oncologist can ease this fear. Knowing your baseline numbers helps you trust the monitoring process.
Side effect management is highly individual. While some tolerate TKIs well, others face grade 3 diarrhea that halts treatment. Support groups online offer practical tips, like specific diets or timing strategies for taking anti-diarrheal meds. Don’t hesitate to lean on community knowledge-it’s invaluable.
What is the difference between HER2-positive and HER2-low breast cancer?
HER2-positive means the cancer cells have a lot of HER2 protein (IHC 3+ or IHC 2+/FISH positive). HER2-low means there is very little HER2 (IHC 1+ or IHC 2+/FISH negative). Historically, HER2-low patients didn’t get targeted therapy. Now, drugs like T-DXd (Enhertu) work for HER2-low metastatic breast cancer, expanding treatment options significantly.
Can HER2-targeted therapies cure breast cancer?
In early-stage disease, yes. Many patients achieve a complete pathological response (no cancer left after neoadjuvant therapy) and remain cancer-free long-term. In metastatic disease, targeted therapies control the disease for years, turning it into a chronic condition rather than a terminal one, though a cure is less common.
Why do I need heart monitoring during treatment?
Drugs like trastuzumab can cause cardiotoxicity, weakening the heart muscle. This isn’t always reversible. Regular echocardiograms monitor your left ventricular ejection fraction (LVEF). If it drops below a certain threshold, your doctor may pause treatment to protect your heart health.
Which drug is best for brain metastases?
Tucatinib (Tukysa) is currently the most effective option. Unlike large antibody drugs, tucatinib is a small molecule that crosses the blood-brain barrier easily. Clinical trials show it significantly improves survival and progression-free survival for patients with HER2-positive brain metastases.
How do I manage diarrhea from TKIs?
Prevention is key. Start taking loperamide (Imodium) before you even feel symptoms if your doctor recommends it. At the first sign of loose stool, take 4mg, then 2mg after each subsequent loose stool, up to 16mg/day. Stay hydrated, eat low-fiber foods, and avoid spicy or fatty meals. Contact your doctor if it persists beyond 24 hours.
Are antibody-drug conjugates safer than chemotherapy?
They are more targeted, meaning less damage to healthy cells, so you avoid classic chemo side effects like hair loss and severe nausea. However, they have unique risks. T-DXd, for example, carries a risk of interstitial lung disease, which requires vigilant monitoring. They aren't "safer" in every way, just different.
What happens if my cancer develops resistance to HER2 therapies?
Resistance is a known challenge. Doctors switch to a different class of drug. If you were on trastuzumab, you might move to T-DM1 or T-DXd. If you were on an ADC, you might try a TKI like tucatinib. Clinical trials are also exploring bispecific antibodies and immunotherapy combinations to overcome resistance.


